Üsküdar Üniversitesi held a press conference to provide information about rare diseases, especially SMA (Spinal Muscular Atrophy), which has been frequently on the agenda recently. Vice Rector Prof. Dr. Muhsin Konuk, emphasizing the importance of conducting domestic studies on rare diseases, including SMA, in our country, stated that our country has the human resources and infrastructure to carry out significant work in this field, and added that Üsküdar Üniversitesi, as TRGENMER, has started studies in this area.
Content
Üsküdar University Starts Studies for the Treatment of SMA and Rare Diseases!
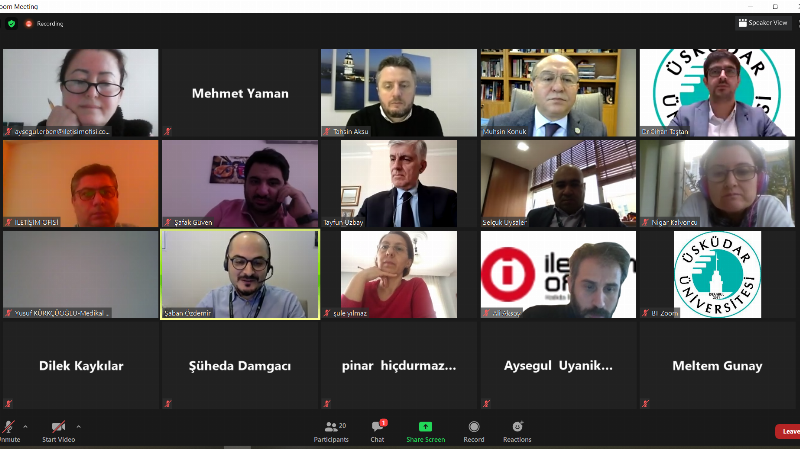
Rector's Advisor Prof. Dr. Tayfun Uzbay, stating that SMA is in the rare diseases group, said, “It is seen with a frequency of 1 in 10,000 worldwide. We observe that this frequency is slightly higher in Turkey. The reason for this, as with all other rare diseases, is consanguineous marriages in our country. Consanguineous marriages also have a significant impact on this disease.”
Lecturer Dr. Cihan Taştan stated that as Üsküdar University TRGENMER, they have started studies to produce similar and cheaper drugs in a national drug concept, to be used in the treatment of rare diseases, especially SMA. He added, “We believe that we will produce these drugs with higher added value and domestically. Therefore, we have completed the preparation of an infrastructure that will conduct R&D and subsequently ÜR-GE (Product Development & Engineering) studies on rare and genetic diseases.”
The press conference, attended by Üsküdar University Vice Rector, Head of the Molecular Biology and Genetics (English) Department Prof. Dr. Muhsin Konuk, Üsküdar University Rector's Advisor, Head of the Internal Medicine Department, Faculty of Medicine Prof. Dr. Tayfun Uzbay, and Director of Üsküdar University's Transgenic Cell Technologies and Epigenetic Application and Research Center (TRGENMER) Lecturer Dr. Cihan Taştan, was held online within the scope of pandemic measures.
Prof. Dr. Tayfun Uzbay: “Consanguineous Marriages Trigger SMA Disease”
Üsküdar University Rector's Advisor, Head of the Internal Medicine Department, Faculty of Medicine Prof. Dr. Tayfun Uzbay, provided information about the methods used in SMA treatment.
Head Prof. Dr. Tayfun Uzbay stated that Spinal Muscular Atrophy (SMA) is a disease characterized by the inability of striated muscles to function properly and a lack of mobility. He said, “It is a condition characterized by the insufficiency, inadequate activity, or insufficient presence of the neurons that control these muscles, which we call motor neurons. The reason for this is the deficiency or mutation of a very important gene called SMN1, which is crucial for providing movement, causing it not to function correctly or fulfill its role. It is part of the rare diseases group. It is seen with a frequency of 1 in 10,000 worldwide. We observe that this frequency is slightly higher in Turkey. The reason for this, as with all other rare diseases, is consanguineous marriages in our country. Consanguineous marriages also have a significant impact on this disease.”
Prof. Dr. Tayfun Uzbay: “SMA Type 1 Negatively Affects Children”
Prof. Dr. Tayfun Uzbay emphasized that, as with all other rare diseases, it is important to investigate and determine whether there is a problem with this gene before marriage and starting a family. He said, “Unfortunately, the disease progresses in a destructive and fatal manner. There are various subtypes. The most discussed ones are type 1, type 2, and type 3. Type 1 usually progresses rapidly in children in the early stages and leads to the child's loss of life. Type 2 and type 3 patients can live a little longer with good care and other supportive treatments that improve their quality of life. There is also type 0, which is never discussed. This is a much more aggressive type of SMA that takes away the baby's chance of survival while still in the womb.”
Prof. Dr. Tayfun Uzbay: “Ministry of Health Supports Treatment”
Head Prof. Dr. Tayfun Uzbay also touched upon the treatment methods for SMA disease. He said, “Currently, there are three drugs for this disease worldwide. The first is a drug we call Nusinersen Sodium. A treatment course of Nusinersen Sodium, which we refer to by its generic name rather than its trade name, costs around 70,000 euros. These are expensive treatments. However, this drug is available in our country. It is the oldest among the three drugs and is covered by the Ministry of Health. If a person has been diagnosed with SMA and is registered in the ministry's system, the Ministry of Health covers the cost of the drug, ensuring treatment. There is another drug called Risdiplam produced by another pharmaceutical company. It has been approved by both the U.S. Food and Drug Administration and the European Medicines Agency.”
Prof. Dr. Tayfun Uzbay: “Assessments and Findings Are Insufficient”
Prof. Dr. Tayfun Uzbay also made evaluations regarding the drug named Onasemnogene Abeparvovec, which has recently come to the agenda. He said, “This drug is new and directly targets the SMN1 gene. With this drug, we introduce the relevant gene into a virus and deliver it to the body. We are talking about a pharmacological application directly related to gene technology. We can see this as the drug treatment of the future. There are two problems here. A single dose of the drug can be obtained for a very high amount, such as 2 million dollars. Unfortunately, there have been very few studies on the drug so far. Given the devastating course and fatal nature of the disease, drugs can be quickly approved by looking at the benefit-risk ratio. Because it is a devastating and fatal disease, patients and their relatives, with hope, want to test and try every new technology. When we scan all reliable sources regarding this drug, we see that there are nearly 60 articles to date. More than half of those articles can be said to be merely commentary and evaluation studies.”
Prof. Dr. Tayfun Uzbay: “Must Be Administered to Children Under 2 Years Old”
Prof. Dr. Tayfun Uzbay stated that the number of children with SMA is small because it is a rare disease. He said, “There are some results in a very small number of individuals. Of course, it is not possible for us to see the long-term results. Currently, we do not have enough data to demonstrate whether it has superiority over the treatment currently applied in Turkey. We will see this over time. This drug absolutely must be administered to children who have not yet reached 2 years of age. The American Food and Drug Administration (FDA) has approved this drug under that condition. We do not know its effects on other types, such as type 2 and type 3, or in older patients. Since it has received approval under such a condition, there is also a possibility that it may be ineffective.”
Prof. Dr. Tayfun Uzbay: “Risks and Benefits Must Be Clearly Stated to Patients”
“Furthermore, another critical issue is that we do not have any scientific evidence regarding whether taking this drug while continuing Nusinersen treatment would be better or worse,” warned Prof. Dr. Tayfun Uzbay. He continued, “Therefore, there have been some speculations about this drug in Turkey. However, it is beneficial to approach this issue calmly and cautiously. Of course, we cannot ignore patient rights. Just as people have the right to choose their treating physician, accept or refuse treatment, they also have the right to try such newly emerged technologies by taking their risks. However, a proper informed consent process is required here, with risks and benefits clearly explained. These types of patients should definitely be selected through an expert consultation, evaluated and examined by a board including a pediatric neurologist and a geneticist, and perhaps used after determining their suitability for this drug.”
Prof. Dr. Tayfun Uzbay stated that they took it upon themselves to act, and as Üsküdar University, they are conducting studies in the field of treatment for SMA and rare diseases.
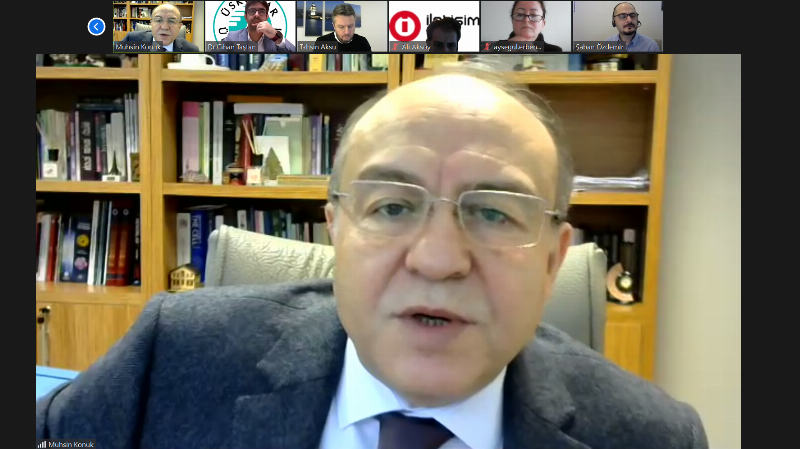
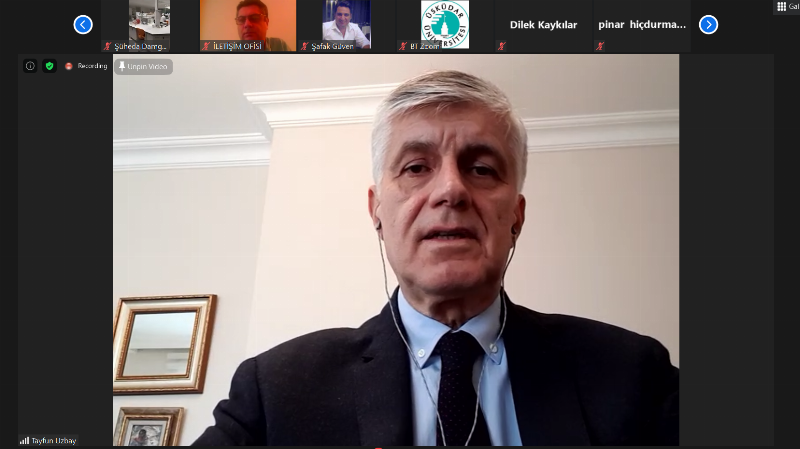
Prof. Dr. Muhsin Konuk: “50% of People with Rare Diseases Are Children”
Üsküdar University Vice Rector, Head of the Molecular Biology and Genetics (English) Department Prof. Dr. Muhsin Konuk stated that SMA disease is among the rare diseases. He said, “Approximately eight thousand rare diseases have been identified today. At least 80% of these rare diseases are genetically transmitted from parents to children. 50% of people affected by rare diseases are children. On average, 30% of these children die before reaching the age of 5. The fundamental reason for this is that rare diseases do not have a real treatment, or rather, almost 95% of them do not have a real treatment.”
Prof. Dr. Muhsin Konuk: “One in 16 People in Turkey Struggles with a Rare Disease”
Prof. Dr. Muhsin Konuk noted that in Europe and developed countries, the concept of rare disease refers to diseases occurring with a frequency of one in two thousand people or less. He added, “Approximately one in 16 people in Turkey continues their life with a rare disease.” Prof. Dr. Muhsin Konuk emphasized that February 29th is specifically commemorated to raise awareness about rare diseases.
Prof. Dr. Muhsin Konuk: “As Üsküdar University, We Established a Center Working on Rare Diseases”
Prof. Dr. Muhsin Konuk stated that in addition to the treatment of these diseases, they also pose a serious wound in society in terms of patient care and certain special costs, and that Üsküdar University is carrying out studies to fulfill its duty in this area. Prof. Dr. Muhsin Konuk said the following: “One in 16 people in Turkey lives with a rare disease, if they can continue to live. Therefore, it is necessary to address these issues and conduct studies to eliminate these diseases. In this context, as Üsküdar University, an institution already focused on behavioral sciences and health and established with that theme, we have established a center aimed at the treatment of these diseases. We can say that genetic treatments for rare diseases are about to begin at this center because all the infrastructure has been established as of now. What is important here is for the country to realize the significance of this work. One of the most important duties of universities is to inform the public correctly, to properly educate society, and to lead society towards positive and good directions with the accumulated knowledge. To find solutions to society's problems, especially considering that genetic treatments, particularly after the development of the CRISPR-Cas9 system for gene editing, have led to this year's Nobel Prize being awarded to the scientists who developed this technology. We have a team that applies this technology very well. Therefore, we held this meeting to inform the public that we are about to start these studies within Üsküdar University in line with today's technology.”
Prof. Dr. Muhsin Konuk: “We Can Establish a Drug System for the Price of a 2 Million Dollar Drug”
Prof. Dr. Muhsin Konuk stated that as Üsküdar University, they have established this infrastructure and, based on it, have started studies related to genetic treatments for diseases, which are currently the most popular form of treatment. He added that the fundamental reason for these difficulties is the scarcity of trained human resources. Emphasizing that our country has the infrastructure and human resources to develop these treatments, he said: “Especially our generation, with almost all of our colleagues sent abroad for PhDs after the 90s returning to our country and provided with certain opportunities, we can establish a drug system in our country in a very short time, perhaps for the price of a single dose of vaccine, or that two million dollar drug. I want to emphasize this because, thankfully, we have the infrastructure and human resources to do it. We wanted to announce this to our colleagues as a preliminary preparation, so that we can correctly inform our public and bring together our existing infrastructure and human brainpower to solve these issues.”
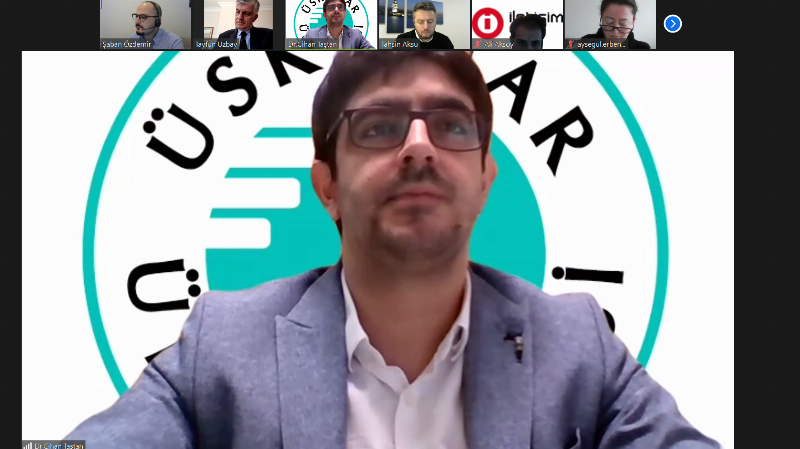
Lecturer Dr. Cihan Taştan: “TRGENMER Works on Genetic Treatments for SMA and Other Rare Diseases”
Lecturer Dr. Cihan Taştan, Director of Üsküdar University Transgenic Cell Technologies and Epigenetic Application and Research Center (TRGENMER), provided information about their work at the center. Lecturer Dr. Cihan Taştan stated that as Üsküdar University, they established TRGENMER to conduct R&D studies on genetic treatments for up to 8,000 rare diseases. He said, “There are 8,000 rare diseases. Among these, we have brought SMA disease to our agenda as one of the most prominent. We currently have a team of 15 people. We are planning R&D studies solely on genetic treatments for SMA disease.”

Provided Information About TRGENMER's Work
Lecturer Dr. Cihan Taştan said, “When we look at the mechanism of the drug currently holding the title of the world's most expensive drug, a functional, working copy of the patient's non-functioning SMN1 gene is directly injected into the vein along with a viral agent. After this injection, the virus containing the SMN1 gene enters the motor neuron cells that cause the symptoms of SMA disease, integrating a functional, working SMN1 gene into the cells. Thus, when we specifically look at the discussed drug, we see that before the age of 2, motor neurons gradually start to improve, and the patient slowly begins to breathe without any assistance. We also have approximately 4 different methods. Currently, two of our methods are similar to the approaches of already approved drugs. One of them is an R&D study we initiated to carry a copy of the SMN1 gene to motor neurons using viral agents.”
Lecturer Dr. Cihan Taştan, also addressing the second of the studies carried out by TRGENMER, said, “On the other hand, our second drug has content named Antisense Oligonucleotide DNA. This is actually a drug approach that I can call genetic treatment-like. Here, the target is a small DNA molecule. This DNA molecule again goes into the motor neurons. But this time, instead of a copy DNA copy of an SMA1 gene, it attaches to the dysfunctional or faulty SMA1 gene, opening the way for it to functionally produce protein again. Of course, this drug is not a long-term drug like the world's most expensive drug or viral genetic therapy, because it does not provide permanent gene expression within the genome or the cell. Therefore, although its price is cheaper than the other, it needs to be taken repeatedly, both in its initial dose and in subsequent doses. We have also started R&D work on DNA-based SMA drugs that incorporate similar approaches by looking at this treatment approach, which is cheaper, has less toxicity (i.e., less harmful to health), and can be practically produced.
On the other hand, we know that CRISPR technologies won the Nobel Prize in 2020. As a scientist who has been working in this field for years, I currently have two different teams using CRISPR technologies. With the different approaches of these two teams, we have also initiated research into whether we can use this in the treatment of SMA disease. Our entire goal is that these technologies are also applicable and treatable for other rare diseases. Since we know that the costs of these drugs are not that expensive, we have started R&D studies as the TRGENMER Center to see if we can produce them similarly and more cheaply within the national drug concept. This allows us to create an environment where, for the first time, a domestic drug for SMA disease, and perhaps later for other rare and genetic diseases, can be produced, and at the same time, families can access these genetic treatments faster and more cheaply.
Our TRGENMER Center is particularly focused on rare diseases. We also have an international genetic treatment competition. Our treatment competition for rare and genetic diseases is called RaDicHal. It takes place between universities. Because we believe that if our university students, whether studying medicine, bioengineering, or genetics, learn these treatment methods and reach a level where they can develop their R&D, we believe we can produce these drugs with higher added value and domestically. Therefore, we have completed the preparation of an infrastructure that will conduct R&D and subsequently ÜR-GE (Product Development & Engineering) studies on rare and genetic diseases.”
Share
Request a Call
Other News
- TRGENMER Participates in Turkey's Space Vision with 6 Papers
- Prof. Nevzat Tarhan Attended Amasya Days
- “Ruha Şifa-Hüsn-ü Hat Exhibition” opened to visitors in Üsküdar again this year…
- Prof. Nevzat Tarhan: “Purpose-driven life provides lasting happiness”
- Quality transformation in higher education discussed

